Safety & Toxicological Testing for
Cosmetics
At AAL Biosciences, cosmetic safety isn’t a checkbox; it’s the foundation of consumer trust. We evaluate how every ingredient, blend, and finished product interacts with the skin, eyes, and biological systems to ensure that beauty never compromises safety.
Aligned with OECD, ISO, BIS, and global regulatory frameworks, our toxicological testing services empower brands to launch products that are gentle, compliant, and ready for global markets.

Necessity of Safety & Toxicological Testing for Cosmetics
Cosmetic safety testing protects consumers, ensures regulatory compliance, and verifies that every formulation is gentle, stable, and fit for real-world use.
Ensures Skin & Eye Safety
Identifies irritation, sensitisation, and ocular risks before products reach consumers.
Validates Ingredient & Formulation Safety
Confirms that raw materials and finished products meet global toxicity and purity standards.
Prevents Microbial & Chemical Contamination
Controls bioburden, endotoxins, heavy metals, and restricted substances to safeguard product integrity.
Supports Regulatory Approvals
Generates data required by EU, FDA, BIS, and global cosmetic regulations for safe market entry.
Protects Brand Credibility
Demonstrates scientific safety verification, building trust with consumers and regulators.
Ensures Long-Term Product Stability
Evaluates preservative performance, phototoxicity, and ingredient interactions across the product lifecycle.
Area of Expertise

Regulatory Compliance Screening
- Analysis of Restricted & Prohibited Substances
Contaminant & Impurity Profiling
- Heavy Metals Analysis
- Residual Solvent Testing

Preservative Efficacy & Microbial Control
- Preservative Efficacy (Challenge Test)
- Microbial Limits & Endotoxin Testing
Dermal Exposure & Absorption
- Dermal Absorption Studies
Genotoxicity & Mutagenicity Testing
- Ames Test, Micronucleus Assay

Systemic & Local Toxicity Studies
- Acute & Repeated Dose Toxicity

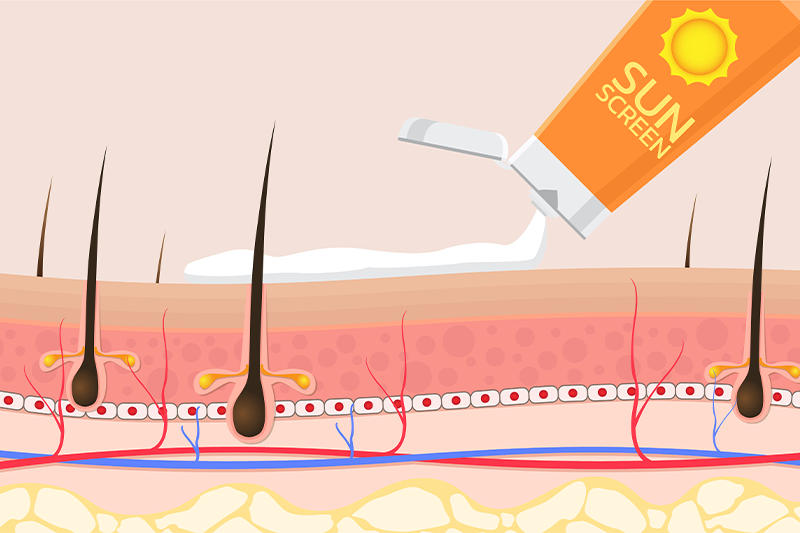
UV-Related Safety Evaluations
- Phototoxicity & Photoallergenicity Tests

Ocular Safety Assessments
- Eye Irritation & Corrosion (BCOP, ICE)
Skin Safety & Sensitisation Studies
- Skin Irritation & Sensitization (OECD 404, LLNA)
